|
|
||
|
|
||
|
How the ‘Oxford’ COVID-19 vaccine became the ‘AstraZeneca’ COVID-19 vaccine At first sight, the Oxford-AstraZeneca COVID-19 vaccine appears to be nothing more than the product of a partnership between a non-profit medical research institute and a wholly profit-oriented pharmaceutical corporation. But as Christopher Garrison shows in the following history of the vaccine, the reality is more complex. 1. Introduction The ‘Oxford/AstraZeneca’ vaccine is one of the world’s leading hopes in the race to end the COVID-19 pandemic. Its history is not as clear, though, as it may first seem. The media reporting about the vaccine tends to focus on either the very small (non-profit, academic) Jenner Institute at Oxford University, where the vaccine was first invented, or the very large (‘Big Pharma’ firm) AstraZeneca, which is now responsible for organising its (non-profit) worldwide development, manufacture and distribution. However, examining the intellectual property (IP) path of the vaccine from invention to manufacture and distribution reveals a more complex picture that involves other important actors (with for-profit perspectives). Mindful of the very large sums of public money being used to support COVID-19 vaccine development, section 2 below will therefore contextualise the respective roles of the Jenner Institute, AstraZeneca and these other actors, so that their share of risk and (potential) reward in the project can be better understood. Section 3 provides comments as well as raising some important questions about what might yet be done better and what lessons can be learnt for the future. 2. History of the ‘Oxford/AstraZeneca’ vaccine 2.1 Oxford University and Oxford University Innovation Ltd The Bayh-Dole Act (1980) was hugely influential in the United States and elsewhere in encouraging universities to commercially exploit the IP they were generating by setting up ‘technology transfer’ offices. Following in those footsteps, Oxford University set up a wholly owned subsidiary company in 1987 to manage the IP generated by its researchers (which was not otherwise owned, for example, by an industrial partner funding the research). This company was originally named Isis Innovation (from the alternative Isis name given to the river Thames as it flows through Oxford) but was renamed Oxford University Innovation (OUI) Ltd in 2016. It is therefore OUI which applies for and manages IP on behalf of Oxford University and its researchers (currently including over 4,000 patents and patent applications). OUI can license the IP out to third parties in return for royalty payments which flow back to OUI and thus Oxford University (and its researchers). Alternatively, OUI can assist the researchers in setting up a spin-out company, supporting it with licences to the IP they generated and initial (‘seed’) funding. Royalty payments may flow back to OUI from the spin-out company and/or OUI may own a stake in it. 2.2 Oxford Sciences Innovation Oxford Sciences Innovation (OSI) is a venture capital firm closely linked with Oxford University which often provides funding to OUI spin-out companies. OSI indicates that, in addition to Oxford University, its diverse shareholders include private- and state-owned investment firms (such as Braavos Capital and Temasek Holdings), venture capital firms (such as Google Ventures (now GV) and Sequoia Heritage), the sovereign wealth fund of Oman, multinational companies (such as Tencent and Fosun Pharma) and the Wellcome Trust, a British charitable body (Figure 1). 2.3 Jenner Institute In 2005, the independent Edward Jenner Institute for Vaccine Research was renamed the Jenner Institute and (in conjunction with the Pirbright Institute, which studies infectious diseases in farm animals) moved to a new institutional home in the Nuffield Department of Medicine at Oxford University. Its work is supported by a charitable body, the Jenner Vaccine Foundation. At present the Jenner Institute hosts 28 research scientists (‘Jenner Investigators’) working on a variety of vaccine-related topics addressing infectious diseases, emerging pathogens and non-communicable diseases. It benefits from close collaboration with several other Oxford University-based entities which permit it to undertake work (on a non-profit basis) at an unusually wide range of points within the vaccine research and development pipeline. For example, the Oxford University Clinical BioManufacturing Facility (CBF) is able to manufacture sufficient quantities of vaccines for at least Phase I/II clinical trials and the Oxford Vaccine Group (OVG), based at the Oxford University Centre for Clinical Vaccinology and Tropical Medicine (CCVTM), is able to undertake clinical trials for vaccines. In recent years the Jenner Investigators have been supported by funding from, for example, the Wellcome Trust, the Gates Foundation, the British Medical Research Council, the British Government Department of Health, the European Commission and the United States National Institutes of Health.
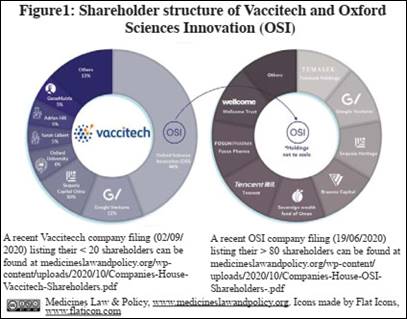
2.4 Vaccitech Ltd Work by two of the leading Jenner Investigators, Sarah Gilbert (Professor of Vaccinology) and Adrian Hill (Professor of Human Genetics, Director of the Jenner Institute), as well as others at the Jenner Institute, led to the development of the Chimpanzee Adenovirus Oxford (ChAdOx) viral vector technology platform. Viral vectors permit artificially selected genetic information to be transported into human (or animal) cells, instructing the cells to manufacture biological material according to that information. Viral vectors can therefore form the basis of a vaccine. If the chosen information corresponds, for example, to a portion of a viral pathogen, when the vector has caused that (harmless but characteristic) portion of the pathogen to be manufactured and introduced to the immune system, the immune system may thereafter be sufficiently primed to respond to any live (dangerous) versions of the whole virus encountered in the future. That response may involve both antibodies and T-cells, the basic building blocks of an immune system response. In order to help further develop the ChAdOx platform, Gilbert and Hill looked to a commercial (for-profit) model. Supported by the legal and business resources of OUI and Ł10 miilion funding from OSI, they therefore founded a spin-out company, Vaccitech, in 2016. OUI had begun to assemble an IP portfolio protecting the ChAdOx viral vector some years before. At least one patent family has been granted in Europe and the United States as well as, for example, China and India, whose 20-year terms expire in 2032. As part of the spin-out process, Vaccitech says that it ‘licensed the needed patents from Oxford University to advance all of the programs’. The terms of this licence, including its scope and exclusivity, are not public. One (investor’s) statement identifies the licence as non-exclusive, at least in the later context of the ChAdOx1 COVID-19 vaccine. Vaccitech says: ‘Our ChAdOx1 vector patent is granted in the US and the EU’ (italics added). In terms of new IP resulting from their development work, Vaccitech says that it ‘further optimises and customises the [ChAdOx] vectors through proprietary promoters and innovative insert design in its Early Development Lab’. Although the scope of the particular package of technology and IP contributed is unclear, Vaccitech now represents that it owns the rights to the ChAdOx platform jointly with Oxford University. Vaccitech has attracted senior management with extensive bio-industry experience, including the present chief executive officer (CEO), Bill Enright, and chief scientific officer (CSO), Thomas Evans. Vaccitech raised Ł20 million in (Series A) venture capital funding (from OSI, GV (US) and Sequoia Capital China) in January 2018, with an $86 million company valuation. In December 2018, Vaccitech raised a further Ł6 million (from GeneMatrix (Korea) and Korea Investment Partners (Korea)). Company records (up to the latest filing on 2 September 2020) indicate that the largest single shareholder of Vaccitech is OSI (~44%) (Figure 1). Against OSI’s initial investment of Ł10 million, at a notional valuation of even $86 million, this 44% stake is worth ~$38 million. Other shareholders include GV (~12%), Sequoia Capital China (~10%), Oxford University (~6%), Sarah Gilbert (~5%), Adrian Hill (~5%), GeneMatrix (~5%), Bill Enright (~3%), Korea Investment Partners (~3%) and Thomas Evans (~1%) (Figure 1). 2.5 The Coalition for Epidemic Preparedness Innovations (CEPI) CEPI was founded in 2017 as a partnership between the governments of Norway and India, the Bill & Melinda Gates Foundation, the Wellcome Trust and the World Economic Forum. Its mission is to ‘stimulate and accelerate the development of vaccines against emerging infectious diseases and enable access to these vaccines for people during outbreaks’. It has now also raised significant funds (over $1 billion) to support its mission from other governments and philanthropic bodies as well as the private sector. The World Health Organisation (WHO), GAVI (formerly the Global Alliance for Vaccines and Immunisations) and CEPI have together launched the COVID-19 Vaccine Global Access (COVAX) initiative with the stated mission of ensuring equitable access to COVID-19 vaccines for all populations.
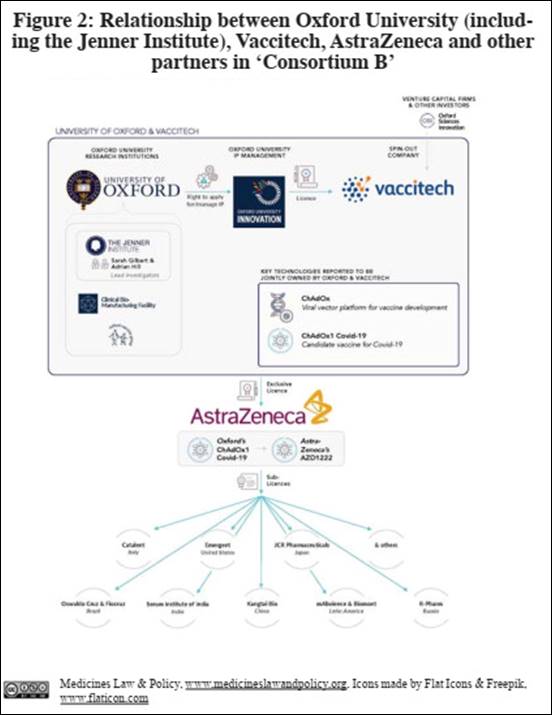
2.6 ChAdOx1 MERS vaccine Middle East Respiratory Syndrome (MERS) is a disease produced by infection with the Middle East Respiratory Syndrome Coronavirus (MERS-CoV). It was first reported in Saudi Arabia in 2012. Coronaviruses are one family of viruses that produce a range of illnesses in humans and animals. They possess a characteristic ‘crown’ of ‘spike protein’ structures which bind with receptors on human (and animal) cells. (The term ‘coronavirus’ stems from ‘corona’ being the Latin word for ‘crown’.) A team at the Jenner Institute (and elsewhere) including Gilbert and Hill developed a MERS vaccine candidate based on their ChAdOx1 viral vector. In this case, the chosen packaged genetic information corresponded to the MERS-CoV ‘spike protein’. In addition to the ChAdOx platform IP, at least one additional patent application was filed by OUI to protect the concept of using the ChAdOx1 viral vector in a MERS vaccine although this now appears to have been abandoned. Vaccitech states that it ‘holds rights to the MERS vaccine ... [and that it has] ... granted certain rights for this vaccine to Oxford to enable development for non-profit public-health use’. Whilst the particular IP on which these rights are based is not specified, Vaccitech therefore reserved the corresponding commercial rights for itself. A Phase I clinical trial undertaken at the Oxford University CCVTM in 2018 (with 24 participants) demonstrated that the ChAdOx1 MERS vaccine was safe and well tolerated and successfully induced an appropriate (antibody and T-cell) immune response in humans. This was an important result in terms of providing preliminary validation for the ChAdOx platform technology. In October 2018, CEPI announced that it would invest $14.6 million to support the further development and manufacture of the ChAdOx1 MERS vaccine (as well as equivalent vaccines for the Nipah virus and Lassa virus) by the Jenner Institute and Janssen Vaccines & Prevention B.V., a subsidiary of Johnson & Johnson. Commenting for the Jenner Institute, Gilbert said: ‘At the Jenner Institute we are strong advocates for the use of proven platform technologies for the rapid and cost-effective development of vaccines against emerging pathogens. In partnership with CEPI and Janssen Vaccines we will use our simian adenoviral vaccine vector ChAdOx1 in preclinical and clinical development of vaccines against MERS, Nipah, and Lassa’ (italics added). Commenting for Vaccitech, however, Evans said: ‘We are delighted that our ChAdOx1 MERS vaccine is being further developed for global public health causes by the strong partnership of Oxford University, Janssen Vaccines & Prevention B.V, and CEPI’ (italics added). The ChAdOx1 MERS vaccine is currently listed by Vaccitech in its portfolio as product ‘VTP-500’ and the project is classed as a ‘co-development’ programme partnered with Oxford University (the Jenner Institute), Janssen and CEPI. A Phase I clinical trial is also now underway in Saudi Arabia, in collaboration with the King Abdullah International Medical Research Centre. 2.7 ChAdOx1 COVID-19 vaccine (Consortium A) Coronavirus Disease 2019 (COVID-19) is a disease produced by infection with another coronavirus, the Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2). It was first reported in China in late 2019. Chinese scientists first publicly disclosed the genetic sequence of SARS-CoV-2 on 10 January 2020. WHO declared the COVID-19 outbreak a public health emergency of international concern (PHEIC) on 30 January 2020 and, given its rapid international spread, declared it a pandemic on 11 March 2020. Given the preliminary validation of the ChAdOx platform in developing a MERS vaccine, it is reasonable to hope that it might likewise be used to develop a COVID-19 vaccine. In this case, the chosen packaged genetic information will instead correspond to the SARS-CoV-2 ‘spike protein’. The Jenner Institute accordingly reports that: ‘A team from the Jenner Institute and Oxford Vaccine Group, led by Prof. Sarah Gilbert, Prof. Andrew Pollard, Prof. Teresa Lambe, Dr Sandy Douglas and Prof. Adrian Hill, started work designing a vaccine [based on the ChAdOx1 viral vector] on Saturday 10th January 2020’ (i.e., the same day that the genetic sequence of SARS-CoV-2 was made publicly available). Vaccitech instead reports that: ‘Oxford University (Oxford) and Vaccitech designed the vaccine in January 2020.’ The ChAdOx1 COVID-19 vaccine candidate is listed in the Vaccitech portfolio as product ‘VTP-900’. As the scale of the COVID-19 outbreak became clear between January and April 2020, the Jenner Institute and Vaccitech looked to a network of partners (here named ‘Consortium A’) to help undertake further work on the ChAdOx1 COVID-19 vaccine candidate and rapidly scale up its manufacture. By early February 2020, it was announced that the CBF was producing the ‘seed stock’ for the ‘Jenner Institute’ ChAdOx1 COVID-19 vaccine candidate and that Advent S.r.l. (Italy) had been contracted to produce the initial doses of the vaccine candidate thereafter to proceed to Phase I/II trials. In partnership with the OVG, these Phase I/II trials (with 1,000 participants) in the UK began in April 2020. By late April, led by Jenner Investigator Sandy Douglas and supported by funding from, for example, UK government research bodies and CEPI, the scaling-up plans had expanded collaboration with partners to include the Vaccine Manufacturing and Innovation Centre (Oxford), Pall Biotech (UK), Cobra Biologics (UK), Halix BV (Netherlands), Merck (Millipore) (US) and the Serum Institute of India (SII). SII is the largest vaccine manufacturer (by doses produced and sold) in the world. Notably, it is privately owned by the Poonawalla family and includes an explicitly philanthropic dimension in its work, permitting it to commit to working as part of Consortium A without having to take into account shareholders’ interests. It is not known whether any of the Consortium A partners contributed any of their own IP to further development or scaling-up. Vaccitech CEO Enright indicated that these ‘unprecedented’ scaling-up plans were focussed on manufacturing in several countries around the world ‘to ensure that no one hoards or unduly prices these products’ and that they should permit ‘millions’ of doses to be manufactured in 2020 and ‘tens to hundreds of millions’ in 2021. Further, reporting on progress on scaling-up made by the ‘Jenner Institute’, an article in the New York Times (27 April 2020) indicated that none of the manufacturing partners had been granted exclusive marketing rights. It explained the absence of a significant (‘Big Pharma’) manufacturing partner in North America in terms of the resistance to offer any firm an exclusive (worldwide) licence. Adrian Hill commented: ‘I personally don’t believe that in a time of pandemic there should be exclusive licences ... So we are asking a lot of them. Nobody is going to make a lot of money off this.’ Adar Poonawala, CEO and owner of SII, also indicated that ‘this is not a time to make money, I think, this is a time to address a huge public health crisis and we should make it affordable and accessible to as many people as possible, especially in the under-privileged countries who need it the most’. Although no explicit commitments were announced, it therefore seems that Consortium A was likely intended to operate on a non-exclusive and low- or non-profit basis. 2.8 ChAdOx1 COVID-19 vaccine (Consortium B) A substantial change in strategy regarding ‘exclusivity’ occurred shortly afterwards. Bill Gates reportedly urged Gilbert and Hill to partner with a ‘Big Pharma’ firm ‘and as a CEPI founder he had leverage’. The UK is home to two ‘Big Pharma’ firms. GlaxoSmithKline (GSK) has a substantial portfolio of vaccines including a COVID-19 vaccine candidate in joint development with Sanofi. By contrast, AstraZeneca has much less prior involvement with vaccines and had no pre-existing COVID-19 vaccine candidate. Despite being reportedly reluctant to do so, it seems that Gilbert and Hill were nevertheless persuaded of the advantages of a tie-up with AstraZeneca by, for example, Sir John Bell (Regius Professor of Medicine, Oxford University and, perhaps not coincidentally, ex-member of the AstraZeneca Scientific Advisory Board) and Sir Mene Pangalos (Executive Vice-President AstraZeneca, Biopharmaceuticals R&D). It seems that AstraZeneca’s organisational scale and reach were regarded as being able to make a potentially vital contribution to further scaling up plans for the ChAdOx1 COVID-19 vaccine candidate. ‘Oxford University’ (the Jenner Institute) and Vaccitech therefore announced on 30 April that an outline agreement had been reached with AstraZeneca, with detailed terms to be negotiated in the following weeks. Vaccitech being a party to this agreement is consistent with their sharing joint ownership of the ChAdOx platform with Oxford University. Making use of its ‘global development, manufacturing and distribution capabilities’, AstraZeneca committed to working with ‘global partners on the international distribution of the vaccine, particularly working to make it available and accessible for low and medium income countries’. This announcement did not mention ‘exclusivity’ and the detailed terms of the agreement are not public. An open letter from various civil society groups to the Jenner Institute sought further clarification of the terms but received no response. AstraZeneca elsewhere stated, however, that ‘AstraZeneca has entered into an exclusive licensing agreement with the University of Oxford for the global development, production and supply of the University’s potential COVID-19 vaccine candidate...’ (italics added). Vaccitech CEO Enright commented that Vaccitech were ‘delighted to facilitate this extensive collaboration’ (italics added). The ChAdOx1 COVID-19 vaccine candidate development is now classed by Vaccitech as an ‘out-licensed’ (rather than ‘co-development’) programme, partnered with Oxford University (the Jenner Institute) and AstraZeneca. The relevant IP covered in the agreement could include, for example, the patents owned by OUI (and licensed to Vaccitech), additional know-how generated by Vaccitech and the clinical trial data generated with the OVG. It is not known whether AstraZeneca will contribute any of its own IP to further development or scaling-up. AstraZeneca refers to the ChAdOx1 COVID-19 vaccine candidate as ‘AZD1222’. The 30 April announcement of the partnership with AstraZeneca specified that the partners will work ‘for the duration of the coronavirus pandemic, with only the costs of production and distribution being covered’. AstraZeneca has reportedly offered to allow at least the US government access to its accounts to verify that this is the case. Once the COVID-19 pandemic is over, however, Oxford University (the Jenner Institute), Vaccitech and AstraZeneca would presumably be free to move to a for-profit exploitation of any successful ChAdOx1 COVID-19 vaccine. It is not specified how the ending of the pandemic will be determined but this must presumably require a formal announcement by WHO. The agreement between the Jenner Institute (Oxford University), Vaccitech and AstraZeneca indicates that any such royalties received by Oxford University will be reinvested directly back in medical research, ‘including a new Pandemic Preparedness and Vaccine Research Centre’, to be developed in conjunction with AstraZeneca. AstraZeneca has subsequently entered into manufacturing and distribution agreements with countries and companies around the world. Much of this activity has to take place ‘at risk’, that is to say, before it is known whether or not the ChAdOx1 COVID-19 vaccine candidate will be successful. It has, however, been supported, and therefore substantially de-risked, by huge sums of public money pledged by governments in return for binding supply agreements of particular numbers of doses. These countries include the United States ($1.2 billion, 300 million doses), the European Union (€330 million, 300 million doses with an option for 100 million more), China (undisclosed sum, 300 million doses), Japan (undisclosed sum, 120 million doses), Brazil ($360m, 100 million doses) and the United Kingdom (Ł65.5 million, 100 million doses). In addition, these agreements have reportedly also included some indemnification for AstraZeneca against future product liability claims arising from the use of the ChAdOx1 COVID-19 vaccine. In Europe, this product liability indemnification is reportedly contingent on AstraZeneca delivering the vaccine at a non-profit price. The companies with which AstraZeneca have reached agreement (in addition to those ‘inherited’ from the Consortium A network) include: Catalent (Italy), Emergent (United States), JCR Pharmaceuticals (plus Daiichi Sankyo, KM Biologics and Meiji Seika Pharma) (Japan), Kangtai Bio (China), Laboratorios Biomont (Mexico), mAbxience (Insud Pharma) (Argentina), Novasep (Belgium), the Oswaldo Cruz Foundation/Fiocruz (Brazil), Oxford Biomedica (United Kingdom), R-Pharm (Russia), SK Bioscience (South Korea) and Symbiosis (United Kingdom). This expanded network of partners is here named ‘Consortium B’ (Figure 2). Consistent with AstraZeneca having been granted exclusive rights, these agreements reportedly include AstraZeneca sub-granting exclusive rights for particular territories. Some agreements reportedly also include a transfer of know-how from AstraZeneca (it is unknown whether this know-how originates with Oxford University and/or Vaccitech or AstraZeneca, or whether there is any obligation for licensees to share any improvements in that know-how back with AstraZeneca or other partners in Consortium B). The appropriate sharing, for example, of clinical trial data for regulatory approval purposes is no doubt also provided for. The agreement with mAbxience and Laboratorios Biomont includes supply of 400 million doses to countries in Latin America. The agreement with R-Pharm includes supply of an undisclosed number of doses to Russia and the ex-Soviet Republics of the Commonwealth of Independent States as well as countries in the Middle East. AstraZeneca’s commitment to ‘working to make [the vaccine] available and accessible for low and medium income countries’ nevertheless seems to rely to a great extent on its agreement with the Serum Institute of India. This includes the supply of 1 billion doses to low- and middle-income countries, including India (of which 400 million are to be supplied by the end of 2020) and, supported by $750 million from CEPI and GAVI, the supply of an additional 300 million doses to the COVAX initiative. SII has given the name ‘Covishield’ to the ChAdOx1 COVID-19 vaccine candidate. However, in the light of the temporary export ban on pharmaceuticals and active pharmaceutical ingredients which the Indian government enacted earlier in the COVID-19 pandemic, SII’s Poonawalla warned that there was no certainty that a similar export ban for the vaccine might not again be put in place. Whilst it is true that the Indian Prime Minister Narendra Modi recently said that ‘India’s vaccine production and delivery capacity will be used to help all humanity in fighting this crisis’, the warning is given particular force by India presently having the fastest-rising COVID-19 caseload of any country. Following the report of a positive outcome to the Phase I/II clinical trials, inducing an appropriate (antibody and T-cell) immune response in humans, Phase II/III clinical trials are now underway in the UK (10,000 participants), Brazil (5,000), South Africa (2,000) and the US (30,000). SII has also launched an independent Phase II/III clinical trial for Covishield (1,600 participants). These clinical trials were paused at the beginning of September due to a (second) suspected adverse event but, at the time of writing, have now resumed in at least the UK, Brazil and India. In sum, if the ChAdOx1 COVID-19 vaccine is successful, AstraZeneca has recently indicated (30 August 2020) that Consortium B is now expected to be able to supply a worldwide total of ‘towards’ three billion doses from 2020 into 2021. 3. Comments 3.1 Balancing public (Oxford University) and private (Vaccitech, OSI etc) interests Rather than being a simple story of a non-profit academic research institute handing over its vaccine candidate to a ‘Big Pharma’ firm, section 2 above illustrates that there are a number of other non- and for-profit parties involved, including Vaccitech and OSI. It is unsurprising that the lines between the Jenner Institute (Oxford University) and Vaccitech are blurred given that Sarah Gilbert and Adrian Hill are both leading investigators in the former as well as co-founders (and ~5% shareholders) of the latter. They and their colleagues at the Jenner Institute (and Vaccitech) deserve a great deal of credit for developing the ChAdOx platform technology. If Vaccitech is indeed the joint owner of the ChAdOx platform technology (section 2.4), however, it seems unarguable that these blurred lines could cause some concern on the part of public funders as to where the balance of risk and reward lies between the two. For example, even if a public funder were to support the development of a particular ChAdOx-based vaccine by the Jenner Institute on a non-profit public health basis, not only might ancillary for-profit markets be reserved for Vaccitech but the successful development of any ChAdOx-based vaccine would further validate the platform technology, benefitting Vaccitech as joint owners with Oxford University. In turn, this will benefit Vaccitech’s shareholders, such as OSI (with its 44% stake in the company), which will, in turn, benefit OSI’s own shareholders, and so on (Figure 1). The ChAdOx1 MERS vaccine provides a concrete example: ‘CEPI will fund the vaccine for public health use. The commercial rights to the vaccine are held by Vaccitech...’ A successful ChAdOx1 COVID-19 vaccine would provide a considerably more important example (see, for example, an article in the Wall Street Journal (2 August 2020): ‘If Oxford’s COVID-19 Vaccine Succeeds, Layers of Private Investors Could Profit’). Of course, private investors in Vaccitech are risking their money in expectation of an attractive return on their investment and, no doubt, the relationship between the Jenner Institute and Vaccitech is regarded as mutually beneficial. Nevertheless, caution is appropriate if the funding of public aims is likely to spill over unduly to private gains. 3.2 Hierarchy of rights (and post-pandemic royalties) In the case of the ChAdOx1 MERS vaccine candidate, Vaccitech chose to divide rights into a non-profit portion, which it granted to the Jenner Institute (Oxford University), and a for-profit portion, which it retained (section 2.6). In the case of the ChAdOx1 COVID-19 vaccine candidate, Vaccitech instead appears to have joined with Oxford University to grant (worldwide) exclusive rights to AstraZeneca, with AstraZeneca then splitting those rights by territory and granting them to relevant partners in Consortium B (section 2.8). Although the Consortium B partners have forgone profit making for the duration of the COVID-19 pandemic, royalty sums could thereafter flow back up this chain of licensees if the vaccine proves a success: from the Consortium B partners to AstraZeneca and from AstraZeneca to Vaccitech and Oxford University (Figure 2). Although Oxford University would use any such royalties to support further non-profit research (section 2.8), they would presumably just represent commercial income for Vaccitech, AstraZeneca and their other partners in Consortium B. This underlines that the ending of the pandemic must be determined on public health grounds through formal announcement by WHO, rather than being determined by commercial considerations.
3.3 AstraZeneca’s assessment of the balance of risks/costs and potential rewards AstraZeneca’s decision to become involved in this initiative (as with each of its Consortium B partners) will have been the result of assessing the balance of its risks and costs with the potential for reward if it succeeds. It is true that AstraZeneca is a very large (‘Big Pharma’) company, with a current market capitalisation of ~$140 billion and a huge portfolio of products, and so can much better afford to take on a project of this unprecedented scale than could, for example, a much smaller pharmaceutical firm with a single product. Further, though, several factors mitigate the risks and costs. As AstraZeneca has explicitly recognised, the ChAdOx technology platform has already been partially validated in the context of MERS (section 2.6) and the Phase I/II results for the COVID-19 vaccine have already been positive (section 2.8). In this respect, it is very different from, for example, the radically new self-amplifying mRNA vaccine technology being developed by Imperial College. Sarah Gilbert was reported in April 2020 as being ‘80% confident’ or alternatively ‘very optimistic’ that the vaccine would work. The costs of developing, manufacturing and distributing the vaccine have also been underwritten with very large sums of public money, now amounting to billions of dollars, and some indemnification appears to have been widely agreed against future product liability claims (section 2.8). If the vaccine is successful, the potential rewards for AstraZeneca and their partners in Consortium B beyond post-pandemic royalties are considerable. Perhaps the most obvious, although difficult to quantify, is the reputational boost associated with delivering a vaccine that will help to overcome the COVID-19 pandemic. Indeed, investment analysts are already praising AstraZeneca CEO Pascal Soriot’s shrewd approach: Andrew Baum (Citibank) commented that ‘Frankly, it’s been a textbook example of optimal public relations and government affairs strategy.’ Again, if the vaccine is successful, AstraZeneca may also be encouraged to build on its experience and deepen its relationship with the Jenner Institute and Vaccitech in order to develop other vaccines using the ChAdOx technology platform. It has been suggested that, if it did so, it could become ‘the go-to pharma firm for future outbreaks’. More broadly, as Vaccitech’s present product portfolio demonstrates, the ChAdOx platform technology is contemplated for use in vaccine applications far beyond new infectious diseases. 3.4 Public health strengths and weaknesses of the Consortium B strategy: increased manufacturing capacity, but overly focussed on high-income countries? From a public health perspective, at least for the immediate future and for the duration of the COVID-19 pandemic, it seems that the pivot from Consortium A to Consortium B has replaced a target of ‘hundreds of millions’ of vaccine doses at a price which is not ‘unduly high’ (section 2.7) with a target of ‘approaching’ three billion vaccine doses at an apparently verifiably non-profit price (section 2.8). In this way, AstraZeneca’s involvement has arguably therefore been very positive. Drilling down, though, it is clear that AstraZeneca’s case-by-case country agreements have initially largely been aimed at serving the populations of the high-income countries (HICs). Such ‘vaccine nationalism’ has been strongly criticised. Further, heavy reliance has been placed on a single (if giant) Indian manufacturer, SII, to deliver the vaccine doses intended for many low- and middle-income countries, including through the COVAX initiative (section 2.8). Bearing in mind that the SII CEO has warned that the exportation of COVID-19 vaccines from India cannot be absolutely guaranteed (section 2.8), it must therefore be important for AstraZeneca to be looking urgently to develop additional manufacturing capacity devoted to low- and middle-income countries and to supporting the COVAX initiative. It is true that the different COVID-19 vaccine ventures are competing for limited manufacturing capacity worldwide. If and where possible, AstraZeneca should therefore be considering supplementary non-exclusive licensing (advantageously through the WHO COVID-19 Technology Access Pool (C-TAP, see box)) and more substantial technology transfer agreements. 3.5 Suggestions to improve the public benefits delivered by Consortium B, including via increased transparency and collaboration Given the huge sums of public money already committed to this project, it is vital that the transparency with which it is operating be improved. For example, AstraZeneca’s reporting of the suspected adverse events in the clinical trials (section 2.8) has been heavily criticised, forcing a response from AstraZeneca. AstraZeneca must also be more forthcoming (beyond governments in the US and Europe) about the cost structure of Consortium B, reassuring the public that it is indeed operating on a non-profit basis, as well as providing important details of its agreements, such as with regard to exclusivity and the scope of the product liability indemnities. It is unacceptable that, as in the Netherlands, negotiations over COVID-19 vaccines have been classified as ‘state secrets’ following the European Commission’s demand to keep vaccine purchase agreements confidential. These concerns about transparency go beyond this particular project too. If the world is aiming for the development of the most effective COVID-19 vaccine possible, then AstraZeneca should arguably also be contemplating pooling the data produced by the clinical trials in an open, collaborative and ‘scientific’ way along with the data produced by other initiatives. Several of them, for example, are also developing vaccine candidates using adenoviruses, including CanSino (China), Gamaleya National Center of Epidemiology and Microbiology (Russia) and Johnson & Johnson (US). The WHO C-TAP (see box) would provide a suitable pooling vehicle. The same will be true of other IP relevant to addressing the COVID-19 pandemic, including, for example, the adenovirus-related manufacturing know-how shared within Consortium B. 3.6 Consortium B as ‘business as usual’ or pointing the way to a new R&D model? Could the scale of the threat posed by the COVID-19 pandemic have encouraged the world to find a superior way of developing and delivering vaccines in time? In common with many other institutions, for example, Oxford University recently pledged to break with ‘business as usual’ in the circumstances of the pandemic and make its COVID-19 IP available through non-exclusive and royalty-free licences (although only for the duration of the pandemic). This is perhaps closer to the approach pursued by Consortium A (and by C-TAP). In the context of Consortium B, however, Oxford University has been criticised for appearing to ‘reverse course’ and agreeing an exclusive deal with AstraZeneca. In fact, the extent to which Oxford University would have been free to act alone is unclear, given its reported joint ownership of the ChAdOx platform technology with Vaccitech (section 2.4). Alternatively, if it could have acted alone, it is unclear what such a split in joint ownership would have involved, in terms of technology and IP. It is also true that the Oxford University pledge is only stated to be a default position and exclusive licensing is seemingly permitted if Oxford University thinks it is necessary to achieve the goal of ‘global deployment at scale’ of the relevant COVID-19-related products. Whatever the particular circumstances, though, and whether Oxford University (and Vaccitech) could have chosen otherwise, exclusive licensing does very much appear to be ‘business as usual’. It is possible, though, to look at the operation of Consortium B during the pandemic from a different and more positive perspective. It could point the way to the future development of a more public-health-focussed research and development (R&D) model. The ‘in-house’ capabilities of Oxford University (including the Jenner Institute, the Clinical BioManufacturing Facility and the Oxford Vaccine Group) already permit a remarkable range of vaccine-related R&D to be carried out (section 2.3). The Jenner Institute (and Vaccitech) did eventually reach out to third-party firms in the private sector regarding their ChAdOx1 COVID-19 vaccine candidate, but in order to scale up production (section 2.7). While AstraZeneca will be contributing significant resources to Consortium B, its most important contribution arguably lies in using its organisational reach and scale to synchronise many more third-party firms’ manufacturing and distribution capacities around the world to enable even further scaling-up. Further, Consortium B is not relying on patents and other IP rights to reap huge profits during the pandemic to fund (further) vaccine development. Instead, the huge sums of public money committed have underwritten the costs of development, manufacture and distribution by each of the Consortium B members, substantially ‘de-risking’ it, helping to ensure that any successful vaccine will be delivered as affordably as possible to as wide a population as possible, to help bring an end to the pandemic. (Again, in Europe, the associated product liability indemnification is reportedly contingent on AstraZeneca delivering the vaccine at a non-profit price, whereas it is instead ‘business as usual’ with no such indemnification for, for example, Sanofi’s for-profit vaccine.) Notwithstanding the involvement of AstraZeneca, a ‘Big Pharma’ firm, the pandemic phase of this project can therefore perhaps be regarded as illustrating at least some aspects of a new de-linked R&D model, where the costs of funding medical R&D and delivering the new products developed are not recovered through monopolistic market pricing. Of course, if this particular project is a success, future analysis will have to judge whether the level of public investment committed was justifiable. Further, since that investment has not seemingly come with adequate post-pandemic strings attached, the ‘business as usual’ model (where patents and other IP rights can be used to try to reap huge profits) might well eventually be expected to reassert itself. Nevertheless, the operation of Consortium B during the pandemic may perhaps be a forerunner pointing to the development of new R&D coalitions and funding models that could serve humanity long beyond the COVID-19 pandemic. Christopher Garrison, MA LLM MA, is a legal adviser with over 20 years of experience on intellectual property issues. He became a consultant legal adviser to Médecins Sans Frontičres’ Campaign for Access to Essential Medicines in 2001. Subsequently he has also consulted on access to medicines, medical R&D and public/private collaboration issues for, for example, the World Health Organisation, the Bill & Melinda Gates Foundation, the Pharmaceutical R&D Policy Project at the London School of Economics, and the Medicines Patent Pool. The above was first published as a briefing paper by Medicines Law & Policy under a Creative Commons licence (CC BY-NC-ND 4.0) and is reprinted here with permission. Medicines Law & Policy brings together legal and policy experts in the field of access to medicines, international law and public health to provide analysis, best practice models and other information that can be used by governments, non-governmental organisations, product development initiatives, funding agencies, UN agencies and others working to ensure the availability of effective, safe and affordable medicines for all. The briefing paper was prepared by Christopher Garrison with the assistance of other members of Medicines Law & Policy: Dr Ellen ’t Hoen, Pascale Boulet and Dr Katrina Perehudoff (review and comments) and Kaitlin Mara (review and comments, infographics and layout). Special thanks are also due to Aaron Blomme (additional input on infographics) and Diarmaid MacDonald (responding to a request regarding the civil society letter (section 2.8)).
*Third World Resurgence No. 345/346, 2020, pp 48-56 |
||
|
|
||